SCIENTIFIC HIGHLIGHTS
Molecular and immunologic breakthroughs are transforming the management of thoracic cancer, although advances have not been as marked for malignant pleural mesothelioma where pathologic diagnosis has been essentially limited to three histologic subtypes. In order to go towards a more multidisciplinary approach for the histologic classification of MPM, we participated in a multidisciplinary group (pathologists, molecular biologists, surgeons, radiologists, and oncologists), sponsored by EURACAN/IASLC to critically review the current classification and issue recommendations. These multidisciplinary recommendations for pathology classification and application will allow more informative pathologic reporting and potential risk stratification, to support clinical practice, research investigation and clinical trials.
While generating the multi-omic data for our own series of MPMs, we started by reanalysing the publicly available data (Bueno et al. Nat Genet. 2016; TCGA Cancer Discov. 2019) with no assumption of discreteness, i.e., without assuming the existence of groups, which is the way the other studies analysed the data. This idea came from the fact that already at the histological level a continuum is suggested, with biphasic MPMs having from 10 to 90% or epithelioid and sarcomatoid components. For this, we undertook unsupervised analyses of RNA-sequencing data of 284 MPMs, and we identified a continuum of molecular profiles that explained the prognosis of the disease better than any discrete model. The immune and vascular pathways were the major sources of molecular variation, with strong differences in the expression of immune checkpoints and pro-angiogenic genes; the extrema of this continuum had specific molecular profiles: a “hot” bad-prognosis profile, with high lymphocyte infiltration and high expression of immune checkpoints and pro-angiogenic genes; a “cold” bad-prognosis profile, with low lymphocyte infiltration and high expression of pro-angiogenic genes; and a “VEGFR2+/VISTA+” better-prognosis profile, with high expression of immune checkpoint VISTA and pro-angiogenic gene VEGFR2 (Figure below). We validated the gene expression levels at the protein level for a subset of five selected genes belonging to the immune and vascular pathways, in a subset of 103 samples and replicated the molecular profiles as well as their prognostic value in the replication series of 77 additional independent samples.
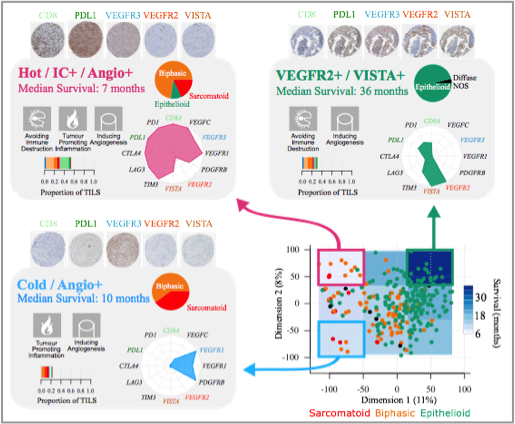
Characteristics of the three MPM transcriptomic profiles. The schematic position of samples harbouring a given profile in the two-dimensional summary (n=284) is represented in the bottom right panel. For each profile, the hallmarks of cancer generally upregulated are indicated by pictograms in the upper left part, the histological type composition is represented by a pie chart in the upper right part, the proportion of tumour infiltrating lymphocytes estimated from the RNA-seq data is represented by a bar plot in the bottom left part, and the expression of representative genes is represented by a radar plot in the bottom right part. Tissue MicroArray IHC staining from the technical validation series, with 0.6 mm core diameter at 5.2× magnification, for the five-gene panel, are presented above each panel (Alcala et al. EBioMedicine 2019).
Through the largest series of whole-genome sequencing data to date, integrated with transcriptomic and epigenomic data using multi-omic factor analysis, we demonstrate that the current WHO classification only accounts for 9% of inter-patient molecular differences. Instead, we propose a novel morpho-molecular classification of MPM based on four dimensions: ploidy, tumor cell morphology, adaptive immune response, and CpG island methylator profile. We show that these four dimensions are complementary, capture major inter-patient molecular differences, and are delimited by extreme phenotypes that, in the case of the interdependent tumor cell morphology and adapted immune response, reflect tumor specialization. These findings unearth the interplay between MPM functional biology and its genomic history, and provide insights into the variations observed in the clinical behavior of MPM patients.
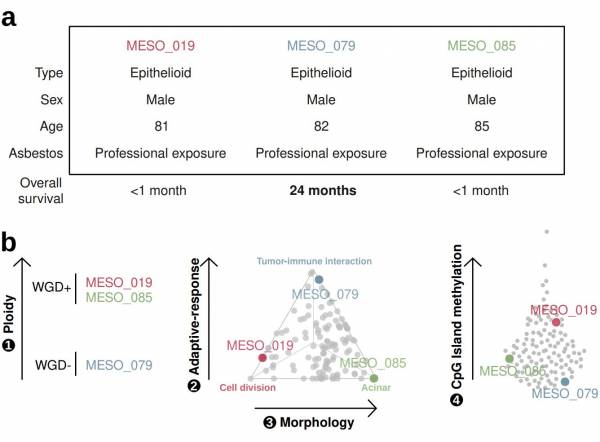
The utility of a four-criteria classification of mesothelioma. Three patients with mesothelioma had similar clinical characteristics yet different outcomes. The three patients have vastly different tumour profiles based on our four-criteria classification. Arrows are directed from low to high values for each criterium, and gray dots represent mesothelioma tumours. (Mangiante et al. Nat Genet 2023).
Our data suggest that MPMs show remarkable morphological intra-tumoral heterogeneity (ITH), both at the cellular composition and the microenvironment, and this has been little studied beyond the histological aspect. Even less is known about how the morphological ITH translates into molecular ITH, despite this might explain the diversity in the response to immunotherapy and anti-angiogenic drugs. We have shown previously that the prognosis of MPM is best explained by a continuum of molecular profiles, with strong differences in tumor immune microenvironment across samples, and that these immune gradients are associated with specific molecular features of the tumor. Studying these interactions is becoming possible with the emergence of single-cell sequencing technologies, which allow to characterize the different cell populations within a tumor, rather than simply the average signal with traditional bulk sequencing. The MESOMICS2 project aims to: (1) build a comprehensive reference molecular map of MPM at the single-cell resolution, (2) quantify and characterize the amount of molecular ITH; and (3) identify predictive candidate biomarkers for aggressiveness and survival that will eventually also guide the design of future clinical trials. Altogether this research will provide insight in biological mechanisms by disentangling the intricate convolution of the signal from the microenvironment with that of the tumor that could not be resolved until now. This will result in biomarkers with stronger diagnostic, prognostic, and predictive values.
FUNDING
Understanding the spatiotemporal eco-evolutionary interactions in malignant pleural mesothelioma. Worldwide Cancer Research (WCR, UK). Grant 24-0106. Coordinators. Active
Intra-tumour heterogeneity of pleural mesothelioma at the single cell level. Department of Defence (DoD, USA). Coordinators. Active.
Tremplin – ERC Consolidator Grant (T-ERC CoG). Agence National de la Recherche (ANR, France). Coordinators. Active
Transcriptomic characterization of biphasic and sarcomatoid malignant pleural mesotheliomas. La Ligue Contre le Cancer (LNCC, France). Appel d’offres 2020. Coordinators. Completed.
Unveiling the intra-tumour heterogeneity of malignant mesothelioma. La Ligue Contre le Cancer (LNCC, France). Appel d'offres 2017. Coordinators. Completed.
Molecular characterisation of malignant pleural mesothelioma. Institut National Du Cancer (INCa, France). PRT-K16-039. Coordinators. Completed.




