SCIENTIFIC HIGHLIGHTS
It is widely accepted that lung neuroendocrine tumours (NETs) and lung neuroendocrine carcinomas (NECs) are different diseases and not simply a continuum of neoplasms with a common pathogenesis. However, over the past years it has been suggested that in both the lung and the thymus the progression or transition from NET to NEC, possibly through the accumulation of genetic anomalies, might be possible. In line with this hypothesis and through multi-omics factor analysis of 116 pulmonary carcinoids (including 35 atypical), 75 large-cell neuroendocrine carcinomas (LCNEC), and 66 small-cell lung cancers, we have identified a group of atypical carcinoids that we have named “supra-carcinoids” with carcinoid-like morphology yet the molecular and clinical features of the deadly LCNEC, suggesting that the molecular link between lung NETs and NECs might be subtler than initially thought (Figure below). In the same study we identified clinically relevant molecular groups of pulmonary carcinoids (Alcala et al., Nat Commun 2019).
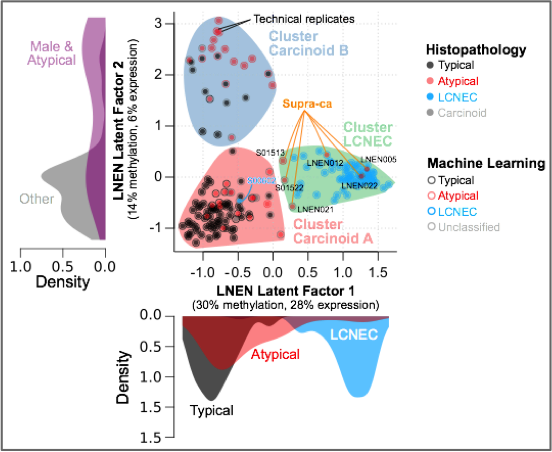
Multi-omics factor analysis (MOFA) of transcriptomes and methylomes of LNEN samples (typical carcinoids, atypical carcinoids, and LCNEC). Point colours correspond to the histopathological types; coloured circles correspond to predictions of histopathological types by a machine learning (ML) algorithm (random forest classifier); filled coloured shapes represent the three molecular clusters identified by consensus clustering. The density of clinical variables that are significantly associated with a latent factor (ANOVA q-value < 0.05) are represented by kernel density plots next to each axis: histopathological type for latent factor 1, sex and histopathological type for latent factor 2.
Another important contribution that we would like to highlight is the generation of a molecular map of lung neuroendocrine neoplasms (Figure below), which provides an interactive way to explore the molecular data and allows easy statistical interrogation, including generating new hypotheses, but also projecting data from studies including fewer samples, (so frequent when working with rare cancers) in order to draw meaningful conclusions (Gabriel et al. Gigascience 2020).
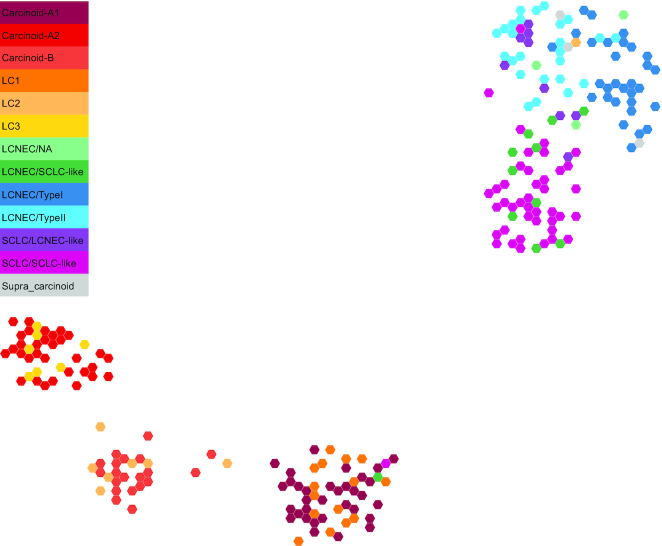
Two-dimensional projection of LNEN transcriptome data using UMAP. The representation was obtained from the TumorMap portal, using the hexagonal grid view, each hexagonal point representing a LNEN sample. Point colours correspond to the molecular clusters defined in the previous publications.
We are currently performing a full molecular characterization of atypical carcinoids and supra-carcinoids (an aggressive group of pulmonary carcinoids that we have recently identified) through a multi-omic integrative analysis (WGS, RNA-seq, and 850k methylation arrays) of 100 primary tumors. The molecular data will be correlated with morphological, epidemiological, and clinical features. Finally, together with T Dayton (in Clever’s lab) we are trying to depict the mechanisms of a possible progression from low to high-aggressive lung neuroendocrine neoplasms by modeling tumor initiation and progression, using state-of-the-art organoid in vitro models.
PUBLICATIONS
Assessment of the current and emerging criteria for the histopathological classification of lung neuroendocrine tumours in the lungNENomics project. Mathian E, Drouet Y, Sexton-Oates, Papotti MG, Pelosi G, Vignaud J-M, Brcic L, Mansuet-Lupo A, Damiola F, Altun C, Berthet J-P, Blanc Fournier C, Brustugun OT, Centonze G, Chalabreysse L, De Montpréville VT, Di Micco CM, Fadel E, Gadot N, Graziano P, Hofman P, Hofman V, Lacomme S, Lund-Iversen M, Mangiante L, Milione M, Muscarella LA, Perrin C, Planchard G, Popper H, Rousseau N, Roz L, Sabella G, Tabone-Eglinger S, Voegele C, Volante M, Walter T, Dingemans A-M, Moonen L, Jan Speel E, Derks J, Girard N, Chen L, Alcala N, Fernandez-Cuesta L, Sylvie Lantuejoul S, Foll M. ESMO Open, 2024. PMID: 38878324.
Clinical management of typical and atypical carcinoids / neuroendocrine tumors in ENETS Centers of Excellence (CoE): Survey from the ENETS lung NET task force. Koumarianou A, Filosso PL, Bodei L, Castano JP, Fernandez-Cuesta L, Deroose CM, Foll M, Dromain C, Reed NS, Caplin M, Capdevila J, Falkerby J, Faggiano A, Frilling A, Grande E, Hicks R, Kasajima A, Kos-Kulda B, Krishna BA, Lim E, Rinke A, Singh S, Thirlwell C, Volante M, Walter T. J. Neuroendocrinol., 2024. PMID: 38754956.
Multi-omic dataset of patient-derived tumor organoids of neuroendocrine neoplasms. Alcala N, Voegele C, Mangiante L, Sexton-Oates A, Clevers H, Fernandez-Cuesta L, Dayton TL, Foll M. GigaScience, Volume 13, 2024. PMID: 38451475.
Druggable Growth Dependencies and Tumor Evolution Analysis in Patient-Derived Organoids of Neuroendocrine Neoplasms from Multiple body sites. Dayton TL, Alcala N, Moonen L, den Hartigh L, Geurts V, Mangiante L, Lap L, Dost AFM, Beumer J, Levy S, van Leeuwaarde RS, Hackeng WM, Samsom K, Voegele C, Sexton-Oates A, Begthel H, Korving J, Hillen L, Brosens LAA, Lantuejoul S, Jaksani S, Kok NF, Hartemink KJ, Klomp HM, Rinkes IHB, Dingemans A, Valk GD, Vriens MR, Buikhuisen W, van den Berg J, Tesselaar M, Derks J, Speel EJ, Foll M, Fernández-Cuesta L, Clevers H. Cancer Cell, 2023. PMID: 38086335. Check the associated Editorial and IARC Press Release.
Spotlight on small-cell lung cancer and other lung neuroendocrine neoplasms. Fernandez-Cuesta L, Sexton-Oates A, Bayat L, Foll M, Lau SCM, Leal T. ASCO Educational Book 2023 PMID: 37229617
Differential Orthopedia Homeobox expression in pulmonary carcinoids is associated with changes in DNA methylation. Moonen L, Mangiante L, Leunissen DJG, Lap LMV, Gabriel A, Hillen LM, Roemen GM, Koch A, van Engeland M, Dingemans AC, Foll M, Alcala N, Fernandez-Cuesta L, Derks JL, Speel EM. Int J Cancer. 2022 PMID: 35076935
A molecular map of lung neuroendocrine neoplasms. Gabriel AAG, Mathian E, Mangiante L, Voegele C, Cahais V, Ghantous A, McKay JD, Alcala N, Fernandez-Cuesta L, Foll M. Gigascience 2020. PMID: 33124659
Accessing and reviewing Controlled Access Data of Rare Cancers. Q&A with Matthieu Foll Gigascience Blog 2020
New molecular classification of large cell neuroendocrine carcinoma and small cell lung carcinoma with potential therapeutic impacts. Lantuejoul S, Fernandez-Cuesta L, Damiola F, Girard N, McLeer A. Transl Lung Cancer Res. 2020. PMID: 33209646
Impact clinique des études moléculaires des tumeurs neuroendocrines pulmonaires. Foll M, Fernandez-Cuesta L. Correspondances en Onco-Thoracique 2020
Molecular studies of lung neuroendocrine neoplasms uncover new concepts and entities. Fernandez-Cuesta L, Foll M. Transl Lung Cancer Res. 2019 PMID: 32038931

Integrative and comparative genomic analyses identify clinically relevant pulmonary carcinoid groups and unveil the supra-carcinoids. Alcala N, Leblay N, Gabriel AAG, Mangiante L, Hervas D, Giffon T, Sertier AS, Ferrari A, Derks J, Ghantous A, Delhomme TM, Chabrier A, Cuenin C, Abedi-Ardekani B, Boland A, Olaso R, Meyer V, Altmuller J, Le Calvez-Kelm F, Durand G, Voegele C, Boyault S, Moonen L, Lemaitre N, Lorimier P, Toffart AC, Soltermann A, Clement JH, Saenger J, Field JK, Brevet M, Blanc-Fournier C, Galateau-Salle F, Le Stang N, Russell PA, Wright G, Sozzi G, Pastorino U, Lacomme S, Vignaud JM, Hofman V, Hofman P, Brustugun OT, Lund-Iversen M, Thomas de Montpreville V, Muscarella LA, Graziano P, Popper H, Stojsic J, Deleuze JF, Herceg Z, Viari A, Nuernberg P, Pelosi G, Dingemans AMC, Milione M, Roz L, Brcic L, Volante M, Papotti MG, Caux C, Sandoval J, Hernandez-Vargas H, Brambilla E, Speel EJM, Girard N, Lantuejoul S, McKay JD, Foll M, Fernandez-Cuesta L. Nat Commun. 2019 PMID: 31431620 – IARC Press Release
A common classification framework for neuroendocrine neoplasms: an International Agency for Research on Cancer (IARC) and World Health Organization (WHO) expert consensus proposal. Rindi G, Klimstra DS, Abedi-Ardekani B, Asa SL, Bosman FT, Brambilla E, Busam KJ, de Krijger RR, Dietel M, El-Naggar AK, Fernandez-Cuesta L, Klöppel G, McCluggage WG, Moch H, Ohgaki H, Rakha EA, Reed NS, Rous BA, Sasano H, Scarpa A, Scoazec JY, Travis WD, Tallini G, Trouillas J, van Krieken JH, Cree IA. Mod Pathol. 2018 PMID: 30140036
Integrative genomic profiling of large-cell neuroendocrine carcinomas reveals distinct subtypes of high-grade neuroendocrine lung tumors. George J, Walter V, Peifer M, Alexandrov LB, Seidel D, Leenders F, Maas L, Müller C, Dahmen I, Delhomme TM, Ardin M, Leblay N, Byrnes G, Sun R, De Reynies A, McLeer-Florin A, Bosco G, Malchers F, Menon R, Altmüller J, Becker C, Nürnberg P, Achter V, Lang U, Schneider PM, Bogus M, Soloway MG, Wilkerson MD, Cun Y, McKay JD, Moro-Sibilot D, Brambilla CG, Lantuejoul S, Lemaitre N, Soltermann A, Weder W, Tischler V, Brustugun OT, Lund-Iversen M, Helland Å, Solberg S, Ansén S, Wright G, Solomon B, Roz L, Pastorino U, Petersen I, Clement JH, Sänger J, Wolf J, Vingron M, Zander T, Perner S, Travis WD, Haas SA, Olivier M, Foll M, Büttner R, Hayes DN, Brambilla E, Fernandez-Cuesta L, Thomas RK. Nat Commun. 2018 PMID: 29535388
New insights into the molecular characteristics of pulmonary carcinoids and large cell neuroendocrine carcinomas, and the impact on their clinical management. Derks JL, Leblay N, Lantuejoul S, Dingemans AC, Speel EM, Fernandez-Cuesta L. J Thorac Oncol. 2018 Review. Erratum in: J Thorac Oncol. 2018 Aug;13(8):1229. PMID: 29454048
Molecular subtypes of pulmonary large-cell neuroendocrine carcinoma predict chemotherapy treatment outcome. Derks JL, Leblay N, Thunnissen E, van Suylen RJ, den Bakker M, Groen HJM, Smit EF, Damhuis R, van den Broek EC, Charbrier A, Foll M, McKay JD, Fernandez-Cuesta L, Speel EM, Dingemans AC; PALGA-Group. Clin Cancer Res. 2018 PMID: 29066508
Frequent mutations in chromatin-remodelling genes in pulmonary carcinoids. Fernandez-Cuesta L, Peifer M, Lu X, Sun R, Ozretić L, Seidal D, Zander T, Leenders F, George J, Müller C, Dahmen I, Pinther B, Bosco G, Konrad K, Altmüller J, Nürnberg P, Achter V, Lang U, Schneider PM, Bogus M, Soltermann A, Brustugun OT, Helland Å, Solberg S, Lund-Iversen M, Ansén S, Stoelben E, Wright GM, Russell P, Wainer Z, Solomon B, Field JK, Hyde R, Davies MP, Heukamp LC, Petersen I, Perner S, Lovly C, Cappuzzo F, Travis WD, Wolf J, Vingron M, Brambilla E, Haas SA, Buettner R, Thomas RK. Nat Commun. 2014 PMID: 24670920
Integrative genome analyses identify key somatic driver mutations of small-cell lung cancer. Peifer M, Fernández-Cuesta L, Sos ML, George J, Seidel D, Kasper LH, Plenker D, Leenders F, Sun R, Zander T, Menon R, Koker M, Dahmen I, Müller C, Di Cerbo V, Schildhaus HU, Altmüller J, Baessmann I, Becker C, de Wilde B, Vandesompele J, Böhm D, Ansén S, Gabler F, Wilkening I, Heynck S, Heuckmann JM, Lu X, Carter SL, Cibulskis K, Banerji S, Getz G, Park KS, Rauh D, Grütter C, Fischer M, Pasqualucci L, Wright G, Wainer Z, Russell P, Petersen I, Chen Y, Stoelben E, Ludwig C, Schnabel P, Hoffmann H, Muley T, Brockmann M, Engel-Riedel W, Muscarella LA, Fazio VM, Groen H, Timens W, Sietsma H, Thunnissen E, Smit E, Heideman DA, Snijders PJ, Cappuzzo F, Ligorio C, Damiani S, Field J, Solberg S, Brustugun OT, Lund-Iversen M, Sänger J, Clement JH, Soltermann A, Moch H, Weder W, Solomon B, Soria JC, Validire P, Besse B, Brambilla E, Brambilla C, Lantuejoul S, Lorimier P, Schneider PM, Hallek M, Pao W, Meyerson M, Sage J, Shendure J, Schneider R, Büttner R, Wolf J, Nürnberg P, Perner S, Heukamp LC, Brindle PK, Haas S, Thomas RK. Nat Genet. 2012 PMID: 22941188
OTHER SCIENTIFIC CONTRIBUTIONS
Since 2020, Lynnette Fernandez-Cuesta and Matthieu Foll are members of the European NEuroendocrine Tumors society (ENETS) – lung NET task force.
Lynnette Fernandez-Cuesta contributed as expert co-author to the Neuroendocrine Tumors chapter of the WHO Classification of Thoracic Tumors, 5th Ed.
Members of the Rare Cancers Genomics Team at IARC have participated as invited speaker, invited discussant, or been selected for oral presentations in the following international renown conferences:
- – 2024 21st Annual ENETS Conference
- – 2023 EMBL Cancer Genomics Conference
- – 2023 NETRF Research Symposium
- – 2023 IASLC World Conference on Lung Cancer
- – 2023 ASCO Annual Meeting
- – 2023 European Congress of Endocrinology
- – 2023 20th Annual ENETS Conference
- – ESMO Sarcoma and Rare Cancers Congress 2023
- – 12th European Meeting on Molecular Diagnostics
- – 2022 IASLC World Conference on Lung Cancer
- – 1st World NET Forum 2022 – Annual ENETS Conference
- – 2021 18th Annual ENETS Conference
- – 2020 NETRF Virtual Research Symposium
- – 2019 Gilles Thomas’ Symposium
- – 2019 31st European Congress of Pathology
- – XV International Symposium GETNE 2019
- – 2019 IASLC World Conference on Lung Cancer
- – 2018 16th Annual ENETS Conference
- – 2018 11th Joint Meeting of the British Division of the IAP and the Pathological Society, together with the NVvP, The Dutch Pathological Society

FUNDING
Reconstructing the evolutionary history of neuroendocrine tumor subtypes. Neuroendocrine Tumor Research Foundation (NETRF, US). 2023 Mentored Award. Coordinators. Active
Reconciling lung carcinoids histopathological and molecular classifications. Neuroendocrine Tumor Research Foundation (NETRF, US). 2022 Investigator Award. Coordinators. Active – IARC Press Release – NETRF 2022 Annual Report
Unveiling the evolutionary processes and molecular pathways underlying the development and progression of lung neuroendocrine neoplasms. Worldwide Cancer Research (WCR, UK). 2020 Grant Round. Coordinators. Active
Comprehensive molecular characterisation of lung supra-carcinoids. Neuroendocrine Tumor Research Foundation (NETRF, US). 2019 Investigator Award. Coordinators. Completed
Genomic characterisation of broncho-pulmonary carcinoids. Institut National Du Cancer (INCa, France). PRT-K17-047. Coordinators. Completed
The Orthopedia Homeobox transcription factor (OTP) in the diagnostics and tumorigenesis of lung carcinoids. KWF Kankerbestrijding (DCS, The Netherlands). Collaborators. Completed
Epigenomic characterisation of lung neuroendocrine tumours. Ligue Contre le Cancer (LNCC, France). Coordinators. Completed
Genomic and transcriptomic characterisation of atypical carcinoids of the lung. National Institutes of Health (NIH, US). R03 CA195253-01. Coordinators. Completed